November 16, 2023 — The Center for Vein Disease at Englewood Health, part of Englewood Health’s Vascular Surgery department, is the first site in the U.S. to utilize a novel medical device for the treatment of varicose veins and their underlying causes.
The ScleroSafe™, a VVT Medical device that recently received clearance from the FDA, offers patients a minimally invasive treatment option for varicose veins without the need for incisions, anesthesia, or thermal heating applied to the tissue.
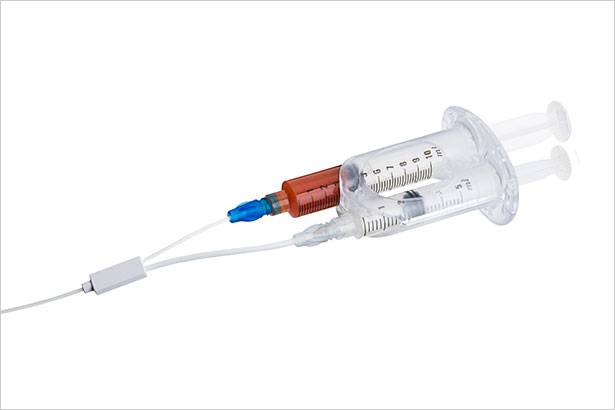
ScleroSafe utilizes a dual syringe technique. During the procedure, one syringe injects a chemical solution used to treat varicose veins, while a second syringe simultaneously removes blood and residual substances from the vein, ensuring unwanted material is not left behind after treatment.
Last week, Steve Elias, MD, Director of the Center for Vein Disease at Englewood Health, performed the first and only ScleroSafe procedures thus far in the U.S.
“The Sclerosafe procedure takes less than 30 minutes, requires only one small needle stick and patients can resume normal activity immediately. We are constantly looking for new treatments to improve our patient’s quality of life. Sclerosafe is the latest addition to our armament of advanced treatment options for vein and vascular conditions,” said Dr. Elias.
“We take great pride in leading the way in the improvement of a varicose vein solution. ScleroSafe holds the promise of transforming varicose vein treatment, providing patients with a safe, effective, and straightforward option,” said Erez Tetro, CEO of VVT Medical.

